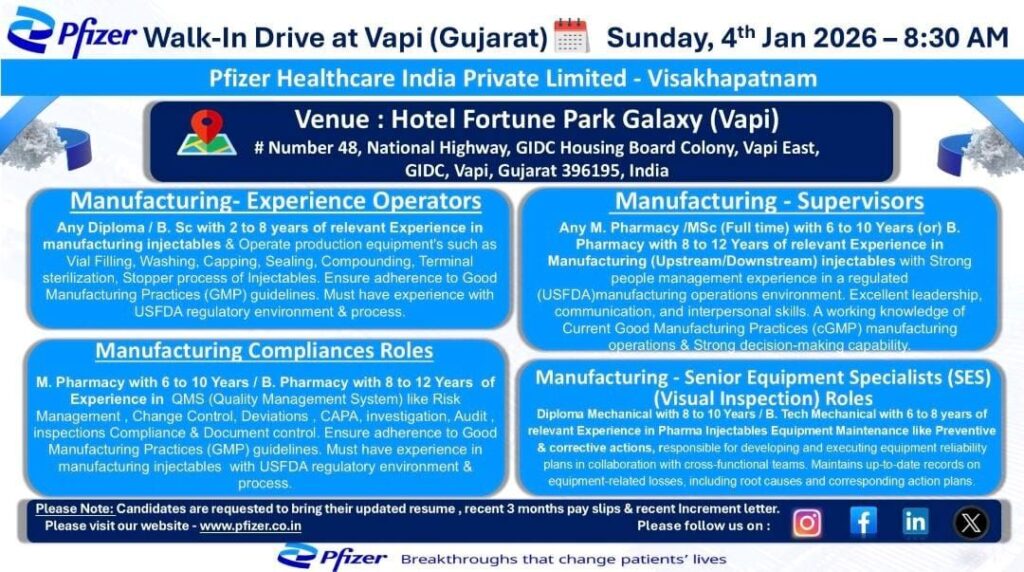
Pfizer Healthcare India Private Limited is conducting a Walk-In Drive at Vapi, Gujarat for experienced professionals in manufacturing, compliance, and equipment roles related to injectable pharmaceutical operations. This is an excellent opportunity to join one of the world’s leading pharmaceutical companies and work in a regulated USFDA & cGMP environment.
Contents
🏢 Company Details
Company Name: Pfizer Healthcare India Private Limited
Business Area: Injectable Manufacturing
Work Location: Visakhapatnam
Walk-In Location: Vapi, Gujarat
Walk-In Interview Details
- Date: Sunday, 4th January 2026
- Time: 8:30 AM onwards
- Venue:
Hotel Fortune Park Galaxy (Vapi)
#48, National Highway, GIDC Housing Board Colony,
Vapi East, GIDC, Vapi, Gujarat – 396195
Open Positions & Eligibility
1️⃣ Manufacturing – Experience Operators
Qualification:
- Diploma / B.Sc
Experience:
- 2 to 8 years in injectable manufacturing
Key Responsibilities:
- Operation of production equipment such as Vial Filling, Washing, Capping, Sealing, Compounding, Terminal Sterilization
- Stopper processing of injectables
- Compliance with GMP guidelines
- Exposure to USFDA regulatory environment
2️⃣ Manufacturing – Supervisors
Qualification:
- M.Pharmacy / M.Sc (Full-time) with 6–10 years experience
OR - B.Pharmacy with 8–12 years experience
Experience Area:
- Injectable manufacturing (Upstream / Downstream)
Key Skills:
- Strong people management
- Leadership & communication skills
- Working knowledge of cGMP manufacturing operations
- USFDA regulated manufacturing experience
- Decision-making and operational excellence
3️⃣ Manufacturing – Compliance Roles
Qualification:
- M.Pharmacy with 6–10 years experience
OR - B.Pharmacy with 8–12 years experience
Experience Area:
- Quality Management Systems (QMS)
Responsibilities:
- Risk Management, Change Control, Deviations, CAPA
- Investigations, Audit handling, Compliance inspections
- Document control
- GMP compliance in injectable manufacturing
- USFDA regulatory exposure mandatory
4️⃣ Manufacturing – Senior Equipment Specialists (SES)
Role: Visual Inspection / Equipment Maintenance
Qualification:
- Diploma Mechanical with 8–10 years experience
OR - B.Tech Mechanical with 6–8 years experience
Experience Area:
- Injectable equipment maintenance
- Preventive & corrective maintenance
- Equipment reliability planning
- Root cause analysis & action plans
- Cross-functional collaboration
- Equipment documentation & compliance
Documents to Carry
- Updated Resume
- Last 3 months payslips
- Recent Increment Letter
Why Join Pfizer?
- Global pharmaceutical leader
- Strong compliance & quality culture
- Exposure to USFDA regulated operations
- Career growth in injectable manufacturing


 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

