Dr. Reddy’s Laboratories Announces Walk-in Drive for Injectable Manufacturing Unit
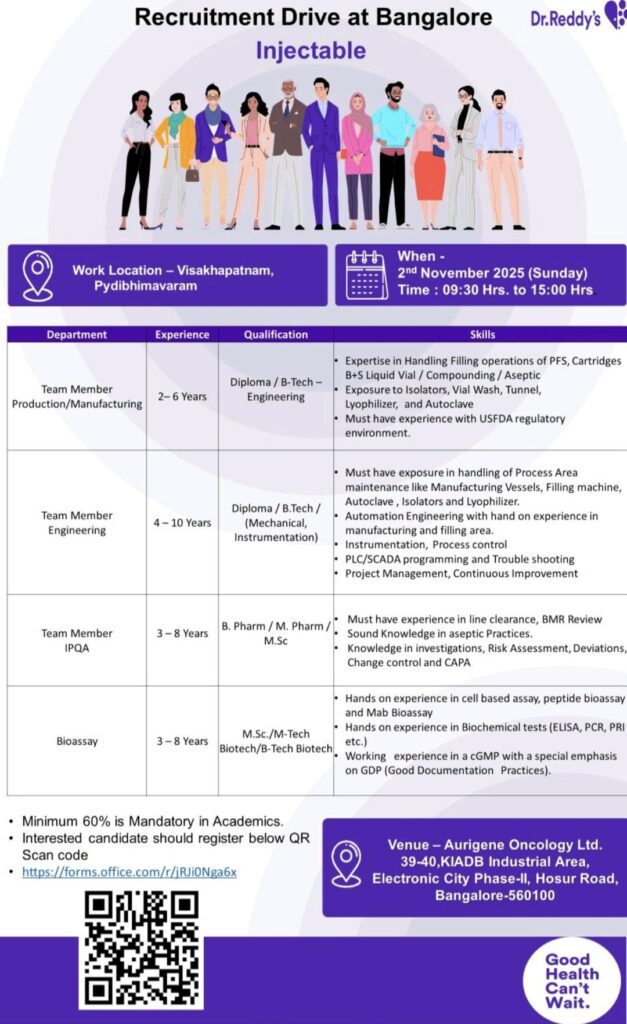
Bangalore, India – In a significant recruitment initiative, Dr. Reddy’s Laboratories, a globally recognized Indian pharmaceutical giant, has announced a walk-in interview drive for its state-of-the-art Injectable manufacturing facility. The event is scheduled for Sunday, November 2, 2025, from 9:30 AM to 3:00 PM in Bangalore.
This drive aims to recruit skilled professionals across various critical functions in the pharmaceutical manufacturing sector, offering opportunities to contribute to the company’s sterile injectables division.
Event Snapshot
Date & Time: 2nd November 2025 (Sunday), 9:30 AM to 3:00 PM
Venue: Aurigene Oncology Ltd.,
39-40, KIADB Industrial Area,
Electronic City Phase-II, Hosur Road,
Bangalore – 560100
Work Location: Selected candidates will be based at the company’s manufacturing units in Visakhapatnam or Pydibhimavaram.
Open Positions and Eligibility
Dr. Reddy’s is looking for experienced candidates in Production, Engineering, Quality Assurance, and Bioassay. A minimum of 60% in academic records is mandatory for all positions.
Here are the detailed openings:
1. Team Member – Production/Manufacturing
Experience: 2 to 6 Years
Qualification: Diploma or B.Tech in Engineering
Required Skills:
· Expertise in handling filling operations for PFS (Pre-filled Syringes), Cartridges, and Liquid Vials (Compounding/Aseptic).
· Hands-on experience with advanced equipment like Isolators, Vial Washers, Tunnels, Lyophilizers, and Autoclaves.
· Familiarity with the USFDA regulatory environment is essential.
2. Team Member – Engineering
Experience: 4 to 10 Years
Qualification: Diploma / B.Tech in Mechanical or Instrumentation
Required Skills:
· Maintenance of Process Area equipment (Manufacturing Vessels, Filling Machines, Autoclaves, etc.).
· Automation Engineering experience in manufacturing/filling areas.
· Proficiency in Instrumentation, Process Control, PLC/SCADA programming, and troubleshooting.
· Knowledge of Project Management and Continuous Improvement methodologies.
3. Team Member – IPQA (In-Process Quality Assurance)
Experience: 3 to 8 Years
Qualification: B.Pharm / M.Pharm / M.Sc
Required Skills:
· Experience in line clearance and Batch Manufacturing Record (BMR) review.
· Sound knowledge of Aseptic Practices.
· Understanding of quality systems: Investigations, Risk Assessment, Deviations, Change Control, and CAPA.
4. Bioassay
Experience: 3 to 8 Years
Qualification: M.Sc./M.Tech/B.Tech in Biotechnology
Required Skills:
· Hands-on experience in cell-based assays, peptide bioassays, and MAb bioassays.
· Proficiency in biochemical tests like ELISA, PCR, and PRI.
· Working experience in a cGMP environment with a strong emphasis on Good Documentation Practices (GDP).
How to Apply?
Interested and eligible candidates can participate through the following methods:
1. Pre-Register Online: Candidates are encouraged to pre-register by scanning the QR code provided on the recruitment poster or by visiting the official registration link: https://forms.office.com/r/RJIONga6x.
2. Walk-in Directly: Attend the interview directly at the specified venue on November 2, 2025, with updated copies of your resume, educational certificates, experience letters, and a passport-sized photograph.
A Premier Opportunity in Pharma
This recruitment drive represents a valuable opportunity for professionals to join the ranks of Dr. Reddy’s, a company renowned for its commitment to quality and global standards. The roles are critical for maintaining the high standards required in sterile injectable manufacturing, with a strong emphasis on USFDA compliance and cutting-edge pharmaceutical technology.
FAQ:
1. What are the key aspects of aseptic processing in injectable manufacturing?
Answer:
Aseptic processing involves the preparation and handling of sterile products where sterilization by terminal means is not possible. Key aspects include:
Maintaining Grade A laminar airflow in critical areas.
Ensuring sterility assurance through environmental monitoring.
Proper gowning and adherence to cleanroom behavior protocols.
Use of isolators and RABS (Restricted Access Barrier Systems).
Validation of aseptic media fills to simulate actual filling operations.
2. What are the main components and functions of a Lyophilizer in sterile manufacturing?
Answer:
A Lyophilizer (Freeze Dryer) is used to remove water from products under vacuum by sublimation.
Main Components:
Condenser: Captures and freezes water vapor.
Chamber: Holds product vials or trays.
Vacuum system: Reduces pressure for sublimation.
Shelves: Provide controlled cooling and heating.
Function:
Ensures product stability for heat-sensitive injectable formulations by converting them into dry powder (lyophilized) form.
3. What is the role of In-Process Quality Assurance (IPQA) in sterile manufacturing?
Answer:
IPQA ensures that quality is built into every stage of production.
Key responsibilities include:
Line clearance before and during production.
Monitoring of aseptic practices and environmental conditions.
Reviewing BMRs (Batch Manufacturing Records) for accuracy.
Verification of sterilization cycles and equipment status.
Control of deviations, ensuring CAPA (Corrective and Preventive Actions) are implemented.
4. Explain the principle and importance of ELISA in Bioassay testing.
Answer:
ELISA (Enzyme-Linked Immunosorbent Assay) is a biochemical technique used to detect and quantify substances such as proteins, peptides, or antibodies.
Principle:
Based on antigen-antibody binding, where an enzyme-linked antibody produces a color change in the presence of its substrate.
Importance:
Used for potency determination, antibody quantification, and bioactivity testing of biologics.
Ensures consistency and efficacy of biopharmaceutical products like monoclonal antibodies and vaccines.
5. How do PLC and SCADA systems support automation in pharmaceutical engineering?
Answer:
PLC (Programmable Logic Controller) and SCADA (Supervisory Control and Data Acquisition) systems automate and monitor manufacturing processes.
Functions:
PLC: Controls machinery (filling lines, autoclaves, sterilizers) through programmed logic.
SCADA: Supervises the process, collects data, and generates alarms for deviations.
Benefits:
Improves process consistency and data integrity.
Reduces human intervention in sterile zones.
Supports 21 CFR Part 11 compliance with electronic data recording.

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

