HOF Pharma Announces Major Recruitment Drive for WHO-GMP Certified Manufacturing Facility in Gujarat
Leading pharmaceutical manufacturer seeks qualified professionals across multiple departments at Sanand and Ahmedabad locations
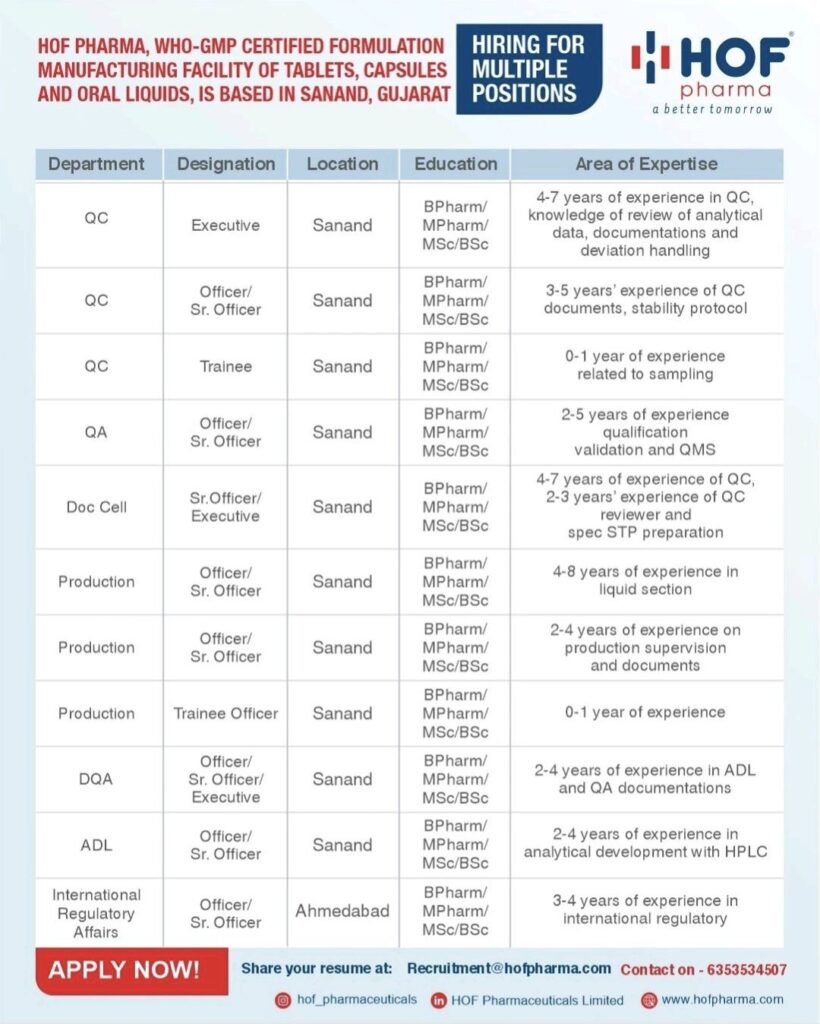
HOF Pharma, a WHO-GMP certified formulation manufacturing facility specializing in tablets, capsules, and oral liquids, has announced an extensive recruitment campaign for various positions across its operations in Gujarat. The company, operating under the tagline “a better tomorrow,” is seeking experienced pharmaceutical professionals to strengthen its workforce at its Sanand and Ahmedabad facilities.
Comprehensive Hiring Initiative
The recruitment drive spans multiple critical departments, offering opportunities for professionals at various career stages, from fresh graduates to seasoned executives with up to eight years of experience.
Quality Control (QC) Department
The QC department is looking to fill positions across three levels. Executive roles require candidates with BPharm/MPharm/MSc/BSc qualifications and 4-7 years of experience in quality control, particularly in reviewing analytical data, documentation, and deviation handling. Officer and Senior Officer positions seek candidates with 3-5 years of experience in QC documents and stability protocols. Entry-level trainee positions are also available for candidates with 0-1 year of experience in sampling-related activities.
Quality Assurance (QA) and Documentation
The QA department is recruiting Officers and Senior Officers with 2-5 years of experience in qualification, validation, and Quality Management Systems (QMS). Additionally, the Documentation Cell is seeking Senior Officers and Executives with 4-7 years of QC experience, including 2-3 years as QC reviewers and expertise in specification and Standard Testing Procedure (STP) preparation.
Production Department
Multiple opportunities are available in the Production department across different experience levels. Senior positions require 4-8 years of experience in liquid section manufacturing, while mid-level roles seek 2-4 years of experience in production supervision and documentation. The department is also offering trainee positions for candidates with 0-1 year of experience.
Specialized Roles
The company is also recruiting for specialized departments including Drug Quality Assurance (DQA), requiring 2-4 years of experience in Approved Drug List (ADL) and QA documentations, and Analytical Development Laboratory (ADL), seeking professionals with 2-4 years of experience in analytical development with High-Performance Liquid Chromatography (HPLC).
For its Ahmedabad location, HOF Pharma is looking for Officers and Senior Officers in International Regulatory Affairs, requiring 3-4 years of experience in international regulatory matters.
How to Apply
Interested candidates meeting the qualification and experience requirements can submit their resumes to Recruitment@hofpharma.com or contact the recruitment team at 6353534507. Additional information about the company and current opportunities can be found on their website at www.hofpharma.com.
The company maintains an active presence on social media platforms, including Instagram (@hof_pharmaceuticals) and LinkedIn (HOF Pharmaceuticals Limited), where candidates can stay updated on company news and career opportunities.
About HOF Pharma
HOF Pharma operates a WHO-GMP certified manufacturing facility in Sanand, Gujarat, focused on formulation manufacturing of tablets, capsules, and oral liquids. The company’s commitment to quality standards and regulatory compliance is reflected in its WHO-GMP certification, positioning it as a reliable pharmaceutical manufacturer in the industry.
FAQ:
1. What qualifications are required for Quality Control (QC) positions at HOF Pharma?
Candidates for QC Executive roles must hold BPharm, MPharm, MSc, or BSc degrees with 4-7 years of experience in analytical data review, documentation, and deviation handling. Officer and Senior Officer positions require 3-5 years of QC experience, while trainee roles accept fresh graduates with 0-1 year of exposure in sampling activities.
2. What experience is needed for Quality Assurance (QA) and Documentation roles?
QA Officers and Senior Officers need 2-5 years of experience in qualification, validation, and Quality Management Systems (QMS). Documentation positions require 4-7 years of QC experience, including 2-3 years as a QC reviewer with expertise in specifications and Standard Testing Procedure (STP) preparation.
3. Are there opportunities in the Production department for fresh graduates?
Yes, HOF Pharma offers trainee positions in Production for candidates with 0-1 year of experience. Mid-level roles require 2-4 years in production supervision, and senior roles need 4-8 years of experience in liquid section manufacturing.
4. What specialized roles are available, and what skills are required?
Specialized departments include Drug Quality Assurance (DQA), requiring 2-4 years of experience in Approved Drug List (ADL) and QA documentation, and the Analytical Development Laboratory (ADL), which seeks professionals with 2-4 years of experience in analytical development using HPLC.
5. How can candidates apply for positions at HOF Pharma?
Interested candidates can send their resumes to Recruitment@hofpharma.com or contact 6353534507. Updated job openings and company information are available on www.hofpharma.com, as well as on Instagram (@hof_pharmaceuticals) and LinkedIn (HOF Pharmaceuticals Limited).

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

