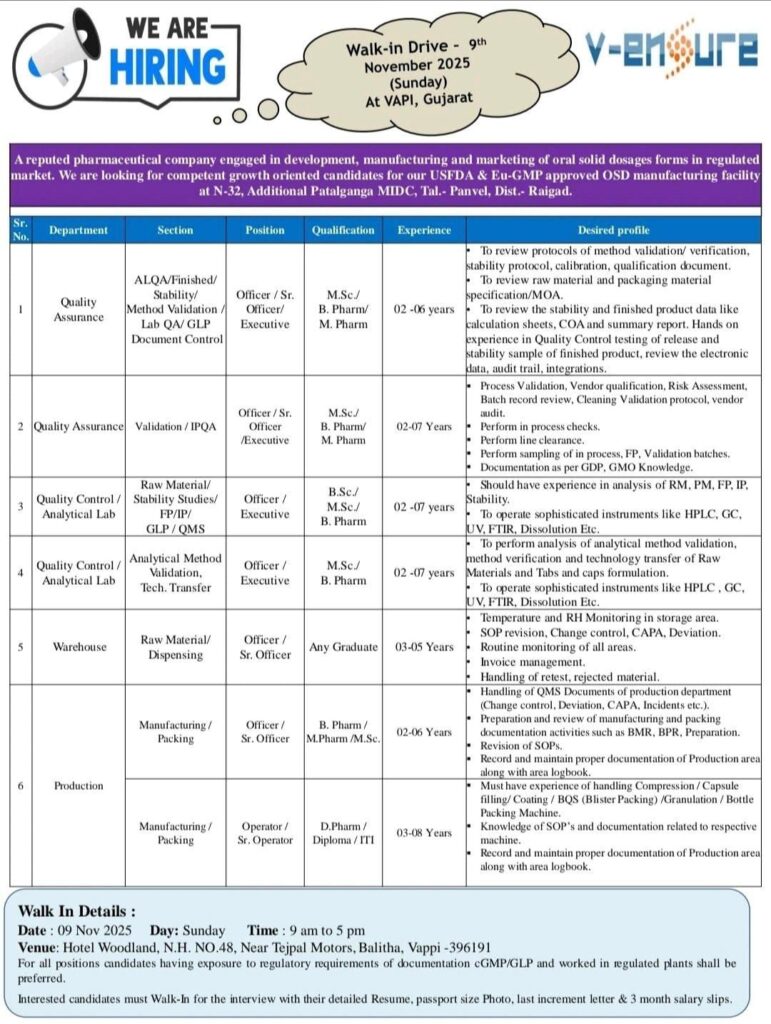
A reputed pharmaceutical company, V-Ensure Pharma, engaged in the development, manufacturing, and marketing of oral solid dosage forms for regulated markets, is conducting a Walk-In Drive for multiple positions. The company is looking for competent and growth-oriented professionals for its USFDA & EU-GMP approved OSD manufacturing facility located at Additional Patalganga MIDC, Tal. Panvel, Dist. Raigad.
Available Departments and Positions
1. Quality Assurance
Sections: ALQA / Finished / Stability / Method Validation / Lab QA / GLP / Document Control
Position: Officer / Sr. Officer / Executive
Qualification: M.Sc. / B.Pharm / M.Pharm
Experience: 02–06 years
Key Responsibilities:
- Review method validation and verification protocols.
- Handle stability protocols and qualification documents.
- Review raw materials and packaging materials specification/MOA.
- Manage finished product data, COA, and summary reports.
- Ensure control of testing and release data, audit trail, and integrations.
2. Quality Assurance (Validation / IPQA)
Position: Officer / Sr. Officer / Executive
Qualification: M.Sc. / B.Pharm / M.Pharm
Experience: 02–07 years
Key Responsibilities:
- Handle validation and vendor qualification activities.
- Oversee cleaning validation, risk assessment, and process validation.
- Manage batch record review and vendor audits.
- Conduct performance checks and ensure compliance with GMP & GDP.
3. Quality Control / Analytical Lab (FP/ IP / GLP / QMS)
Position: Officer / Executive
Qualification: B.Sc. / M.Sc. / B.Pharm
Experience: 02–07 years
Key Responsibilities:
- Operate analytical instruments such as HPLC, GC, UV, and FTIR.
- Perform analytical tests, review results, and handle documentation.
4. Quality Control / Analytical Lab (Analytical Method Validation & Tech Transfer)
Position: Officer / Executive
Qualification: M.Sc. / B.Pharm
Experience: 02–07 years
Key Responsibilities:
- Conduct analytical method validation and verification.
- Support technology transfer of raw materials and finished formulations.
- Operate instruments like HPLC, GC, UV, and FTIR.
5. Warehouse (Raw Material / Dispensing)
Position: Officer / Sr. Officer
Qualification: Any Graduate
Experience: 03–05 years
Key Responsibilities:
- Monitor storage area temperature and RH.
- Handle CAPA, deviation, and SOP revisions.
- Manage invoices, rejected materials, and retests.
6. Manufacturing / Packing
Position: Officer / Sr. Officer
Qualification: B.Pharm / M.Pharm / M.Sc.
Experience: 02–06 years
Key Responsibilities:
- Prepare and review manufacturing and packing documentation.
- Revise SOPs and handle BMR/BPR activities.
- Manage production data and maintain area logbooks.
7. Production
Position: Operator / Sr. Operator
Qualification: D.Pharm / Diploma / ITI
Experience: 03–08 years
Key Responsibilities:
- Operate compression, capsule filling, blister packing, and granulation machines.
- Maintain production documentation and SOPs.
Candidate Requirements
- Should have exposure to regulatory requirements of cGMP / GLP.
- Experience in working with regulated pharmaceutical plants preferred.
- Must carry updated resume, passport-size photo, last increment letter, and 3-month salary slips.
Walk-In Drive Details
📅 Date: 9th November 2025 (Sunday)
🕘 Time: 9:00 AM – 5:00 PM
📍 Venue: Hotel Woodland, N.H. No.48, Near Tejpal Motors, Balitha, Vapi – 396191

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

