USV Private Limited Announces Major Walk-in Drive for Vadodara Pharma Facility
In a significant recruitment initiative, USV Private Limited, a leading name in the Indian pharmaceutical industry, has announced a comprehensive walk-in interview event. The company is seeking to onboard skilled professionals for its state-of-the-art formulations manufacturing facility located in Savli-Manjusar, Vadodara.
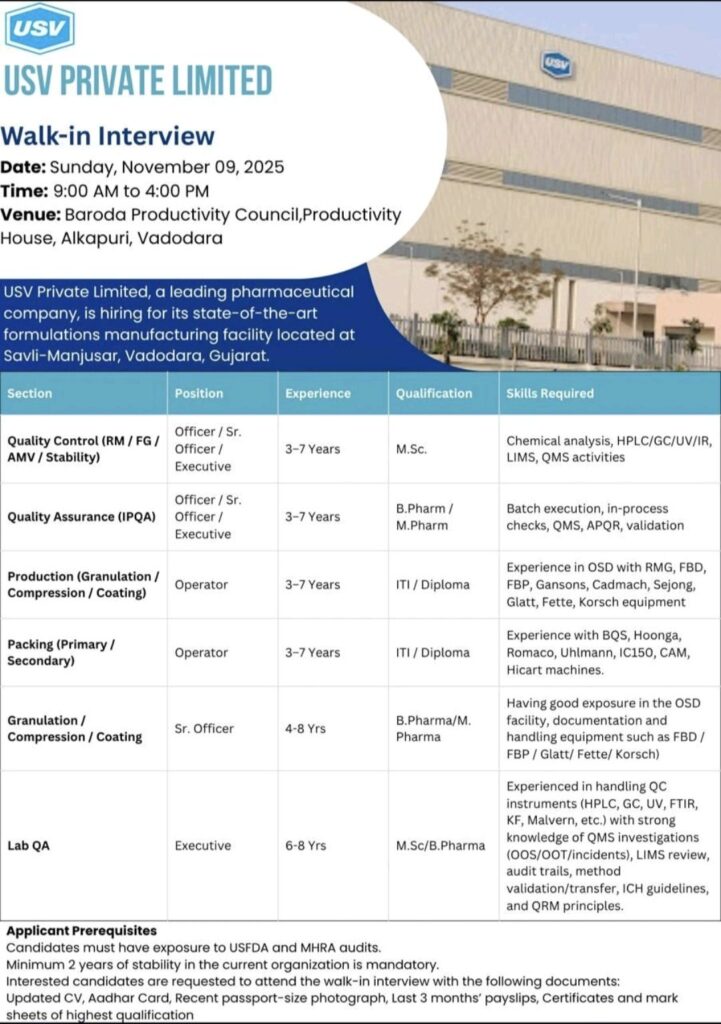
Scheduled for Sunday, November 9, 2025, the day-long drive will be held at the Baroda Productivity Council, Productivity House, Alkapuri, Vadodara, from 9:00 AM to 4:00 PM.
This recruitment drive underscores the company’s expansion and commitment to bolstering its workforce with experienced talent across critical operational verticals.
Open Positions and Eligibility
USV Private Limited has opened vacancies across several departments, targeting candidates with specific technical expertise and qualifications. The available roles are:
1. Quality Control (RM / FG / AMV / Stability)
Positions: Officer / Sr. Officer / Executive
Experience: 3 to 7 Years
Qualification: M.Sc.
Key Skills: Hands-on experience in chemical analysis, and operating instruments like HPLC, GC, UV, and IR. Familiarity with LIMS and QMS activities is essential.
2. Quality Assurance (IPQA)
Positions: Officer / Sr. Officer / Executive
Experience: 3 to 7 Years
Qualification: B.Pharm / M.Pharm
Key Skills: The role requires proficiency in batch execution, conducting in-process checks, and handling QMS, APQR, and validation activities.
3. Production & Packing Departments
Positions: Operator (Production & Packing)
Experience: 3 to 7 Years
Qualification: ITI / Diploma
Key Skills:
· Production: Candidates should have experience in Oral Solid Dosage (OSD) facilities with equipment like RMG, FBD, FBP, Gansons, Cadmach, Sejong, Glatt, Fette, and Korsch.
· Packing: Experience operating BQS, Hoonga, Romaco, Uhlmann, IC150, CAM, and Hicart machines is required.
4. Granulation / Compression / Coating (Sr. Officer)
Position: Sr. Officer
Experience: 4 to 8 Years
Qualification: B.Pharma / M.Pharma
Key Skills: The role demands strong exposure in OSD facility operations, documentation, and handling advanced equipment such as FBD, FBP, Glatt, Fette, and Korsch.
5. Lab QA
Position: Executive
Experience: 6 to 8 Years
Qualification: M.Sc. / B.Pharma
Key Skills: This senior role requires extensive experience in handling QC instruments (HPLC, GC, UV, FTIR, KF, Malvern) and a strong grasp of QMS investigations (OOS/OOT/incidents), LIMS review, audit trails, method validation/transfer, ICH guidelines, and QRM principles.
Mandatory Prerequisites for Applicants
The company has set specific prerequisites to ensure candidate suitability:
· All applicants must have prior exposure to USFDA and MHRA audits.
· A minimum of 2 years of stability in the current organization is mandatory.
Documents to Carry
Interested and eligible candidates are requested to attend the walk-in with the following documents:
· An updated Curriculum Vitae (CV)
· Aadhaar Card for identity proof
· Recent passport-size photograph
· Last 3 months’ payslips
· Certificates and mark sheets of their highest qualification
A Career Opportunity with an Industry Leader
This walk-in interview presents a valuable opportunity for pharmaceutical professionals to join a reputed organization known for its high standards and advanced manufacturing capabilities. USV Private Limited’s facility in Savli-Manjusar is a benchmark for quality and compliance, offering a dynamic environment for career growth
FAQ:
1. What is the difference between OOS (Out of Specification) and OOT (Out of Trend)?
Answer:
OOS (Out of Specification): Refers to test results that fall outside the established acceptance criteria or specification limits.
OOT (Out of Trend): Refers to results that are within specification limits but show a trend shift or abnormal variation compared to historical data.
Both require investigation, but OOS focuses on immediate product quality issues, while OOT helps in trend monitoring and process control.
2. Explain the working principle of HPLC and why it is used in QC labs.
Answer:
HPLC (High-Performance Liquid Chromatography) works on the principle of separation based on the differential partitioning of analytes between a stationary phase and a mobile phase.
The sample mixture is injected into a column packed with stationary phase material.
Components move at different rates depending on their interactions, resulting in separation.
Purpose: It is used for quantitative and qualitative analysis of drugs, impurities, and degradation products in pharmaceutical formulations.
3. What are critical parameters to monitor during granulation and compression in OSD manufacturing?
Answer:
Granulation: Moisture content, binder concentration, mixing time, and particle size distribution.
Compression: Tablet weight, hardness, thickness, disintegration time, and compression force.
Monitoring these ensures content uniformity, flowability, and tablet integrity, which are crucial for consistent product quality.
4. How is Line Clearance performed in IPQA, and why is it important?
Answer:
Line clearance is a documented activity carried out before starting any new batch or product to ensure the area and equipment are clean and free from previous product residues or materials.
Steps:
Check for cleanliness, labels, and materials.
Verify removal of previous batch documents.
Fill and sign line clearance checklist.
Importance: Prevents cross-contamination, mix-ups, and ensures compliance with cGMP guidelines.
5. What are QMS elements you have handled in your previous organization?
Answer:
Typical Quality Management System (QMS) elements include:
Deviation Management
Change Control
CAPA (Corrective and Preventive Action)
OOS/OOT Investigation
Document Control
Internal Audits
Demonstrating knowledge of these shows your understanding of regulatory compliance and quality culture in a pharmaceutical setup

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

