Senores Pharmaceuticals Limited, a rapidly growing pharmaceutical company with a strong global presence, is conducting Walk-In Interviews for multiple positions across R&D, Formulation Development, ADL/ QC, Documentation, and Regulatory Affairs departments. This is an excellent opportunity for experienced pharma professionals to advance their careers with a quality-driven organization.
Contents
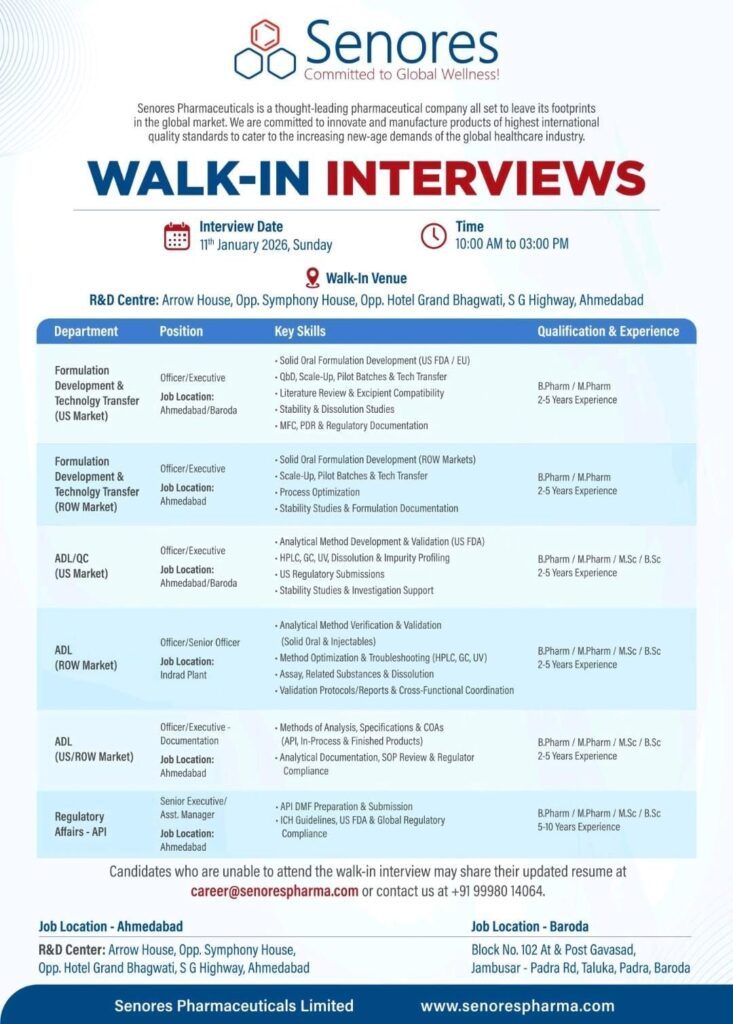
🔹 Current Job Openings1. Formulation Development & Technology Transfer – US Market2. Formulation Development & Technology Transfer – ROW Market3. ADL / QC – US Market4. ADL – ROW Market5. ADL – Documentation (US / ROW Market)6. Regulatory Affairs – APIWalk-In Interview Details📩 Unable to Attend Walk-In?🏭 Job Locations
🔹 Current Job Openings
1. Formulation Development & Technology Transfer – US Market
- Position: Officer / Executive
- Location: Ahmedabad / Baroda
- Key Skills: Solid oral formulation (US FDA/EU), QbD, scale-up, pilot batches, tech transfer, stability & dissolution studies
- Qualification: B.Pharm / M.Pharm
- Experience: 2–5 Years
2. Formulation Development & Technology Transfer – ROW Market
- Position: Officer / Executive
- Location: Ahmedabad
- Key Skills: Solid oral formulation (ROW), scale-up, process optimization, formulation documentation
- Qualification: B.Pharm / M.Pharm
- Experience: 2–5 Years
3. ADL / QC – US Market
- Position: Officer / Executive
- Location: Ahmedabad / Baroda
- Key Skills: Analytical method development & validation, HPLC, GC, UV, impurity profiling, US regulatory submissions
- Qualification: B.Pharm / M.Pharm / M.Sc / B.Sc
- Experience: 2–5 Years
4. ADL – ROW Market
- Position: Officer / Senior Officer
- Location: Indrad Plant
- Key Skills: Method validation, troubleshooting, assay, dissolution, analytical coordination
- Qualification: B.Pharm / M.Pharm / M.Sc / B.Sc
- Experience: 2–5 Years
5. ADL – Documentation (US / ROW Market)
- Position: Officer / Executive – Documentation
- Location: Ahmedabad
- Key Skills: COAs, specifications, analytical documentation, SOP review, regulatory compliance
- Qualification: B.Pharm / M.Pharm / M.Sc / B.Sc
- Experience: 2–5 Years
6. Regulatory Affairs – API
- Position: Senior Executive / Assistant Manager
- Location: Ahmedabad
- Key Skills: API DMF preparation, ICH guidelines, US FDA & global regulatory compliance
- Qualification: B.Pharm / M.Pharm / M.Sc / B.Sc
- Experience: 5–10 Years
Walk-In Interview Details
- Interview Date: 11 January 2026 (Sunday)
- Time: 10:00 AM to 03:00 PM
- Venue:
R&D Centre – Arrow House, Opp. Symphony House,
Opp. Hotel Grand Bhagwati, S G Highway, Ahmedabad.
📩 Unable to Attend Walk-In?
Candidates who cannot attend the walk-in interview may send their updated resume to:
career@senorespharma.com
📞 Contact: +91 99980 14064
🏭 Job Locations
- Ahmedabad: R&D Centre, S G Highway
- Baroda: Jambusar–Padra Road, Padra

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

