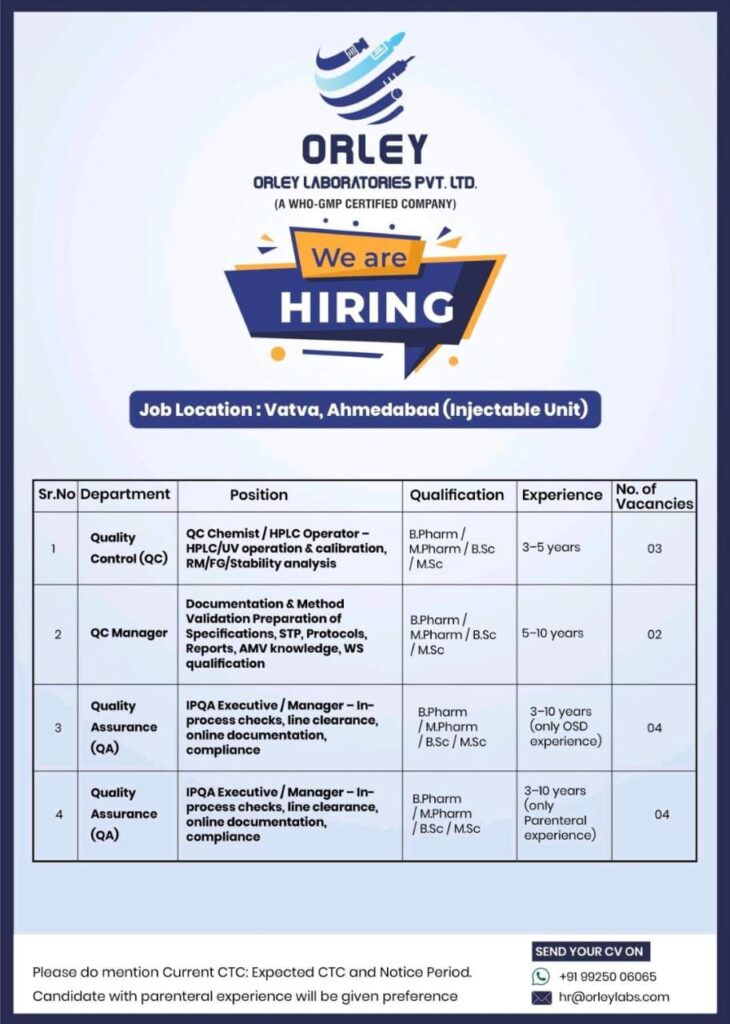
Orley Laboratories Pvt. Ltd., a WHO-GMP certified pharmaceutical company, is inviting applications for various Quality Control (QC) and Quality Assurance (QA) roles at its Injectable Unit in Vatva, Ahmedabad.
📍 Job Location: Vatva, Ahmedabad (Injectable Unit)
Available Positions
🔹 1. Quality Control (QC) – QC Chemist / HPLC Operator
Responsibilities:
- Operation and calibration of HPLC/UV systems
- RM, FG, and stability analysis
Qualification: B.Pharm / M.Pharm / B.Sc / M.Sc
Experience: 3–5 years
Vacancies: 03
🔹 2. QC Manager
Responsibilities:
- Documentation and Method Validation
- Preparation of specifications, STP, protocols, reports
- AMV knowledge and WS qualification
Qualification: B.Pharm / M.Pharm / B.Sc / M.Sc
Experience: 5–10 years
Vacancies: 02
🔹 3. Quality Assurance (QA) – IPQA Executive / Manager (OSD)
Responsibilities:
- In-process checks and line clearance
- Online documentation and compliance monitoring
Qualification: B.Pharm / M.Pharm / B.Sc / M.Sc
Experience: 3–10 years (Only OSD experience)
Vacancies: 04
🔹 4. Quality Assurance (QA) – IPQA Executive / Manager (Parenteral)
Responsibilities:
- In-process checks, line clearance, online documentation
- Regulatory and compliance adherence
Qualification: B.Pharm / M.Pharm / B.Sc / M.Sc
Experience: 3–10 years (Only Parenteral experience)
Vacancies: 04
Preferred Candidate Profile
- Candidates with parenteral experience will be given preference.
- Mention Current CTC, Expected CTC, and Notice Period in your application.
How to Apply
📧 Email your CV: hr@orleylabs.com
📞 Contact: +91 99250 06065
🌟 Why Join Orley Laboratories?
- WHO-GMP Certified Company
- Dynamic & Growth-Oriented Work Culture
- Competitive Salary and Benefits
- Exposure to International Quality Standards

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

