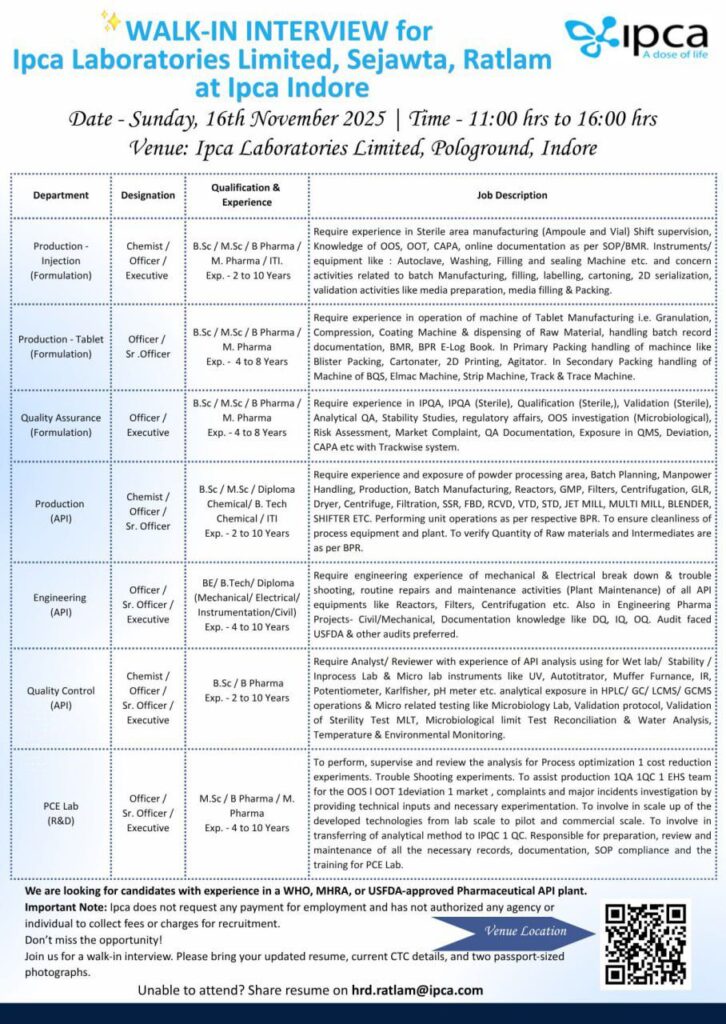
Ipca Laboratories Limited, a leading pharmaceutical company with WHO, MHRA, and USFDA-approved API plants, is conducting a walk-in interview for multiple positions across Production, Quality Assurance, Quality Control, R&D, and Engineering departments. This is an excellent opportunity for professionals seeking a rewarding career in a reputed pharma organization.
Walk-In Interview Details:
- Date: Sunday, 16th November 2025
- Time: 11:00 AM to 4:00 PM
- Venue: Ipca Laboratories Limited, Pologround, Indore
Available Positions:
1. Production (Injection & Tablet Formulation)
Openings for Chemist, Officer, and Executive roles. Candidates should hold a B.Sc., M.Sc., B.Pharm., or M.Pharm. degree with 2 to 10 years of experience. The role involves sterile manufacturing, machine operations like granulation, compression, coating, packing, and working knowledge of GMP documentation such as BMR and BPR. Candidates with hands-on experience in sterile areas and OOS/OOT/CAPA processes will be preferred.
2. Quality Assurance (Formulation)
Openings for Officer and Executive positions for candidates with B.Sc., M.Sc., B.Pharm., or M.Pharm. qualifications and 4 to 8 years of experience. Applicants should have experience in IPQA, Qualification, Validation, Analytical QA, Stability Studies, and exposure to QMS, Deviation, CAPA, and Trackwise systems.
3. Production (API)
Chemist, Officer, and Sr. Officer roles are open for candidates with B.Sc., M.Sc., Diploma, or B.Tech in Chemical or Mechanical fields with 2 to 10 years of experience. Candidates must have experience in process operations, reactors, centrifugation, filtration, and batch manufacturing under GMP standards.
4. Engineering (API)
Openings for Officer, Sr. Officer, and Executive roles for BE/B.Tech/Diploma holders in Mechanical, Electrical, Instrumentation, or Civil Engineering with 4 to 10 years of experience. Candidates must have expertise in plant maintenance, troubleshooting, and managing API plant equipment like reactors, filters, and centrifuges. Knowledge of calibration, IQ/OQ/PQ documentation, and audit compliance is desirable.
5. Quality Control (API)
Ipca is hiring Chemists, Officers, and Executives with B.Sc. or B.Pharm. degrees and 2 to 10 years of experience. Candidates should be experienced in analytical instruments such as HPLC, GC, UV, and Karl Fischer, as well as microbiological and wet lab testing for APIs.
6. PCE Lab (R&D)
Openings for Officer and Sr. Officer positions. Candidates must have M.Sc., B.Pharm., or M.Pharm. with 4 to 10 years of experience. Responsibilities include process optimization, cost reduction studies, OOS investigation, and analytical method development. Experience in lab-scale to pilot-scale process validation is essential.
Important Notes:
- Candidates must have experience in WHO, MHRA, or USFDA-approved pharmaceutical API plants.
- No recruitment fees are required—Ipca does not authorize any agency to collect money for employment.
- Candidates are advised to carry an updated resume, current CTC details, and two passport-sized photographs.
If unable to attend the interview, you can share your resume via email at hrd.ratlam@ipca.com.
Don’t miss this opportunity to be part of Ipca Laboratories.

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

