Ipca Laboratories Ltd., a leading name in the pharmaceutical industry, is conducting a walk-in interview drive for multiple positions across Quality Assurance, IPQA, AQA, and AMV departments. This is a great opportunity for qualified professionals looking to build a rewarding career in one of India’s most trusted pharmaceutical companies.
Contents
Job Openings at Ipca Laboratories Ltd1. Officer / Sr. Officer – Quality Assurance (QMS)2. Officer / Sr. Officer – Quality Assurance (Documentation)3. Officer / Sr. Officer / Executive / Sr. Executive – Quality Assurance (Qualification)4. Officer / Sr. Officer / Executive / Sr. Executive – IPQA (Packing)5. Officer / Sr. Officer / Executive / Sr. Executive – IPQA (Manufacturing)6. Officer / Sr. Officer – AQA7. Officer / Sr. Officer – AMVWhy Join Ipca Laboratories Ltd.?Important Instructions
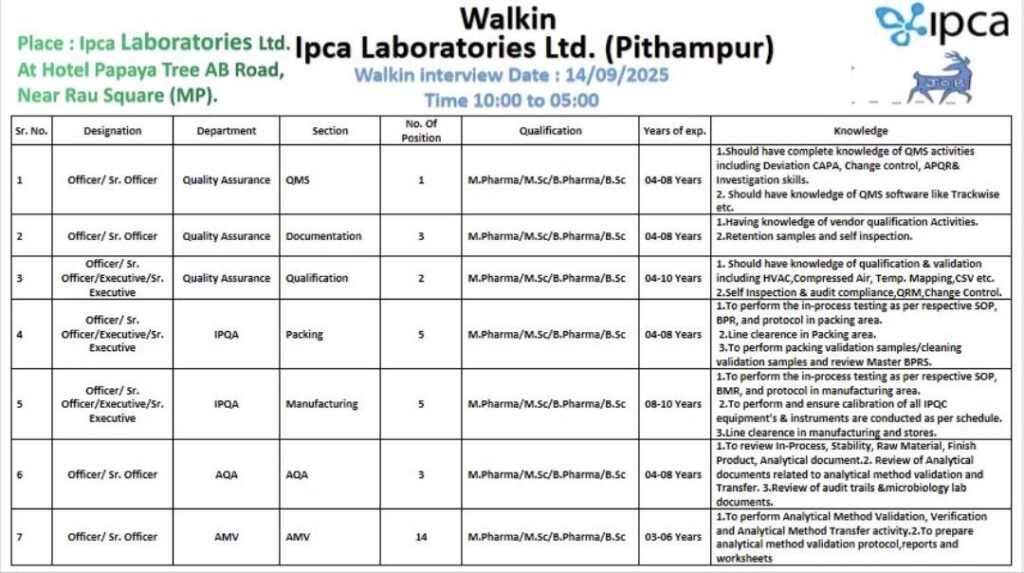
📍 Venue: Hotel Papaya Tree, AB Road, Near Rau Square, Pithampur (MP)
📅 Date: 14th September 2025 (Sunday)
⏰ Time: 10:00 AM – 5:00 PM
Job Openings at Ipca Laboratories Ltd
| Designation | Department | Section | No. of Positions | Qualification | Experience | Key Knowledge / Skills Required |
|---|---|---|---|---|---|---|
| Officer / Sr. Officer | Quality Assurance | QMS | 1 | M.Pharm / M.Sc / B.Pharm / B.Sc | 4–8 years | QMS activities (Deviation, CAPA, Change Control, APRQR, Investigations), QMS software (Trackwise) |
| Officer / Sr. Officer | Quality Assurance | Documentation | 3 | M.Pharm / M.Sc / B.Pharm / B.Sc | 4–8 years | Vendor qualification, retention samples, self-inspection |
| Officer / Sr. Officer / Executive / Sr. Executive | Quality Assurance | Qualification | 2 | M.Pharm / M.Sc / B.Pharm / B.Sc | 4–10 years | Qualification & validation, HVAC, compressed air, temp mapping, CSV, audit compliance |
| Officer / Sr. Officer / Executive / Sr. Executive | IPQA | Packing | 5 | M.Pharm / M.Sc / B.Pharm / B.Sc | 4–8 years | In-process testing, line clearance, packing validation |
| Officer / Sr. Officer / Executive / Sr. Executive | IPQA | Manufacturing | 5 | M.Pharm / M.Sc / B.Pharm / B.Sc | 8–10 years | In-process testing, BMR/BPR compliance, stores & warehouse clearance |
| Officer / Sr. Officer | AQA | AQA | 3 | M.Pharm / M.Sc / B.Pharm / B.Sc | 4–8 years | Analytical method documentation, analytical reports, audits, microbiology knowledge |
| Officer / Sr. Officer | AMV | AMV | 14 | M.Pharm / M.Sc / B.Pharm / B.Sc | 3–6 years | Analytical Method Validation, Protocol Preparation, Reports & Worksheets |
1. Officer / Sr. Officer – Quality Assurance (QMS)
- Positions: 1
- Qualification: M.Pharm / M.Sc / B.Pharm / B.Sc
- Experience: 4–8 years
- Knowledge Required:
- QMS activities: Deviation, CAPA, Change Control, APRQR, Investigations
- QMS software (e.g., Trackwise)
2. Officer / Sr. Officer – Quality Assurance (Documentation)
- Positions: 3
- Qualification: M.Pharm / M.Sc / B.Pharm / B.Sc
- Experience: 4–8 years
- Knowledge Required:
- Vendor qualification activities
- Retention samples and self-inspection
3. Officer / Sr. Officer / Executive / Sr. Executive – Quality Assurance (Qualification)
- Positions: 2
- Qualification: M.Pharm / M.Sc / B.Pharm / B.Sc
- Experience: 4–10 years
- Knowledge Required:
- Qualification & validation (HVAC, Compressed Air, Temp. Mapping, CSV)
- Self-inspection and audit compliance (QRM, Change Control)
4. Officer / Sr. Officer / Executive / Sr. Executive – IPQA (Packing)
- Positions: 5
- Qualification: M.Pharm / M.Sc / B.Pharm / B.Sc
- Experience: 4–8 years
- Knowledge Required:
- In-process testing as per SOP, BPR, and protocols
- Line clearance in packing area
- Packing validation samples, cleaning validation, and Master BPR
5. Officer / Sr. Officer / Executive / Sr. Executive – IPQA (Manufacturing)
- Positions: 5
- Qualification: M.Pharm / M.Sc / B.Pharm / B.Sc
- Experience: 8–10 years
- Knowledge Required:
- In-process testing as per SOP, BMR, and protocols
- Monitoring manufacturing activities
- Line clearance in manufacturing and stores
6. Officer / Sr. Officer – AQA
- Positions: 3
- Qualification: M.Pharm / M.Sc / B.Pharm / B.Sc
- Experience: 4–8 years
- Knowledge Required:
- Analytical method documentation & review
- Analytical reports, raw data, protocol preparation
- Review of audit trails & microbiology
7. Officer / Sr. Officer – AMV
- Positions: 14
- Qualification: M.Pharm / M.Sc / B.Pharm / B.Sc
- Experience: 3–6 years
- Knowledge Required:
- Analytical Method Validation (AMV)
- Analytical method transfer activity
- Preparation of validation protocols, reports, and worksheets
Why Join Ipca Laboratories Ltd.?
- Be part of a leading pharmaceutical organization with global recognition.
- Opportunities across multiple QA and Manufacturing functions.
- Exposure to regulatory compliance, analytical validation, and quality systems.
- Career growth and learning opportunities in a structured, professional environment.
Important Instructions
- Candidates should carry their updated resume, passport-size photographs, and relevant educational/experience certificates.
- Ensure you meet the required qualifications and experience before attending the interview.
Don’t miss this chance to accelerate your career with Ipca Laboratories Ltd. Walk in with confidence and take the next step toward your professional growth!

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

