IPCA Laboratories Ltd. Announces Major Walk-In Recruitment Drive for Pharma Professionals
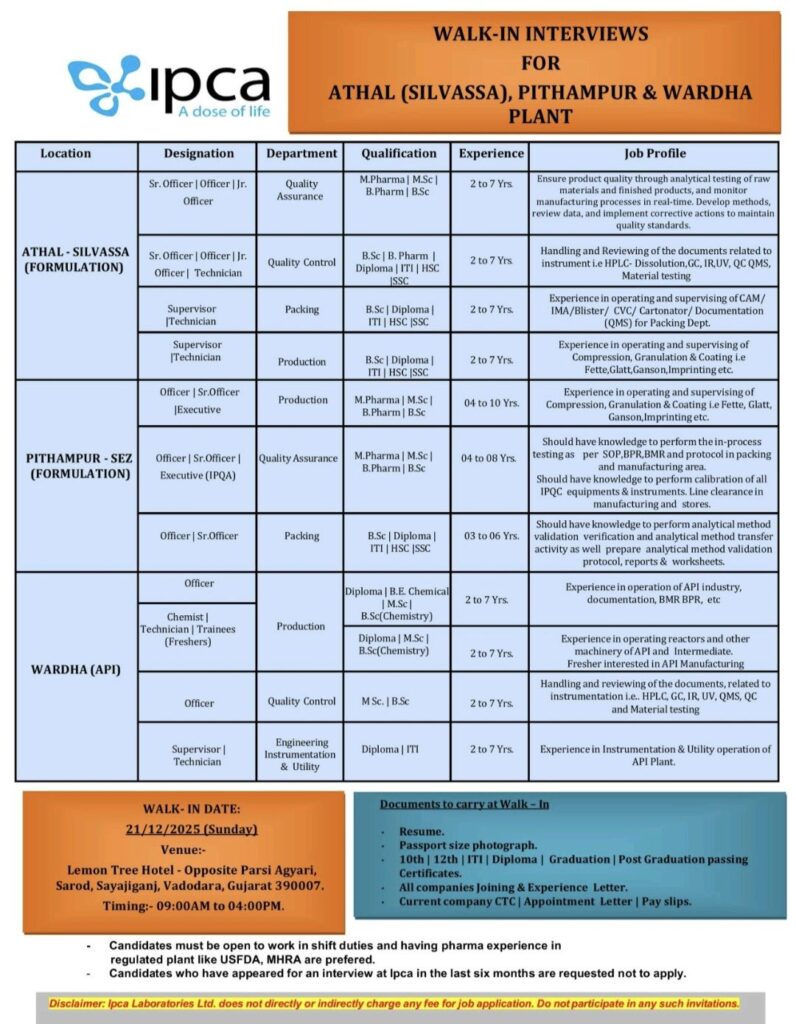
IPCA Laboratories Ltd., a globally recognized Indian pharmaceutical company, is conducting a large-scale walk-in interview drive to hire talented professionals across its key manufacturing facilities. The company is seeking candidates for its formulation plants in Athal (Silvassa) and Pithampur (SEZ), as well as its API (Active Pharmaceutical Ingredient) plant in Wardha. This is an excellent opportunity for experienced individuals and promising freshers to build a career with an industry leader.
Interview Details at a Glance:
Date: Sunday, 21st December 2025
Time: 9:00 AM to 4:00 PM
Venue: Lemon Tree Hotel, Opposite Parsi Agiyari, Sarod, Sayajigunj, Vadodara, Gujarat – 390007.
Available Opportunities by Location:
1. Athal, Silvassa (Formulation Plant)
This unit has openings in Quality Assurance,Quality Control, Production, and Packing departments.
Roles: Senior Officer, Officer, Junior Officer, Supervisor, Technician.
Qualification: M.Pharm, B.Pharm, M.Sc, B.Sc, Diploma, ITI, HSC/SSC.
Experience: 2 to 7 years.
Desired Skills: Hands-on experience in analytical testing (HPLC, GC, UV), QMS documentation, supervising compression/granulation/coating operations (Fette, Glatt machines), and packing line operations (Blister, CVC, Cartonator).
2. Pithampur – SEZ (Formulation Plant)
Openings are available for mid-level roles in Production and Quality Assurance.
Roles: Officer, Senior Officer, Executive.
Qualification: M.Pharm, B.Pharm, M.Sc, B.Sc.
Experience: 3 to 10 years.
Desired Skills: Strong knowledge of in-process quality control (IPQC), SOP/BPR/BMR compliance, equipment calibration, and analytical method validation/transfer activities.
3. Wardha (API Plant)
The API plant invites applications for Production,Quality Control, and Engineering roles, including openings for freshers.
Roles: Officer, Chemist, Technician, Supervisor, Trainees (Freshers).
Qualification: M.Sc (Chemistry), B.Sc (Chemistry), B.E. Chemical, Diploma, ITI.
Experience: 2 to 7 years (Freshers can apply for Trainee positions).
Desired Skills: Experience in API manufacturing operations, reactor handling, documentation (BMR/BPR), and instrumentation & utility operations.
Mandatory Documents to Bring:
Interested candidates must carry the following documents for the interview:
Updated resume.
Passport-sized photograph.
Original and copies of all educational certificates (10th, 12th, Graduation, Post-Graduation, Diploma, ITI).
Previous and current company experience letters, appointment letters, and latest pay slips.
Current CTC proof.
Key Preferences & Notes:
· Candidates must be willing to work in shift rotations.
· Prior experience in plants regulated by agencies like USFDA or MHRA will be a significant advantage.
· Candidates who have attended an interview with IPCA Laboratories in the past six months are requested not to apply.
Why Consider a Career with IPCA?
IPCA Laboratories Ltd. offers a dynamic work environment, exposure to international quality standards, and structured career growth paths in the pharmaceutical sector. This walk-in drive presents a direct opportunity to join a company committed to quality healthcare and employee development.
Disclaimer: IPCA Laboratories Ltd. does not charge any fee at any stage of the recruitment process. Candidates are cautioned against any fraudulent communication asking for payment.
FAQ:
1. What technical knowledge is expected for Production roles in Formulation and API plants?
Candidates should have practical knowledge of manufacturing processes such as granulation, compression, coating, and packing for formulation units, and reactor operations, crystallization, drying, and material handling for API plants. Understanding of BMR/BPR documentation, equipment operation, and adherence to cGMP guidelines is essential.
2. Which analytical instruments and testing techniques should Quality Control candidates be proficient in?
QC professionals are expected to have hands-on experience with HPLC, GC, UV–Visible Spectrophotometers, Dissolution Testers, and wet chemistry techniques. Knowledge of method validation, stability studies, calibration, and data integrity (ALCOA principles) is highly desirable.
3. What Quality Assurance (QA) responsibilities are critical for these roles?
QA candidates should be well-versed in IPQC activities, SOP preparation and review, deviation handling, change control, CAPA, risk assessment, and batch release processes. Familiarity with regulatory audits (USFDA, MHRA, WHO-GMP) and compliance documentation is a strong advantage.
4. Are freshers eligible for technical roles, and what skills are expected from them?
Yes, freshers are eligible mainly for Trainee positions at the API plant (Wardha). They should possess strong theoretical knowledge, basic understanding of GMP, safety practices, chemical processes, and a willingness to learn plant operations, documentation, and regulatory compliance.
5. How important is regulatory exposure in the selection process?
Regulatory exposure is a key technical advantage. Candidates with experience in USFDA, MHRA, EU-GMP, or other international audits will be preferred, as IPCA operates facilities that comply with global quality and regulatory standards.

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

