Location: Ludhiana, Punjab
Company: Indllina Pharmaceuticals
Industry: Pharmaceuticals | WHO-GMP Certified Manufacturing
Indllina Pharmaceuticals, a fast-growing WHO-GMP certified pharmaceutical company based in Ludhiana, is expanding its technical and quality team. The organization is committed to manufacturing excellence, global compliance standards, and continuous innovation. If you are looking to build a rewarding career in a modern, compliant facility, explore the openings below.
Available Vacancies
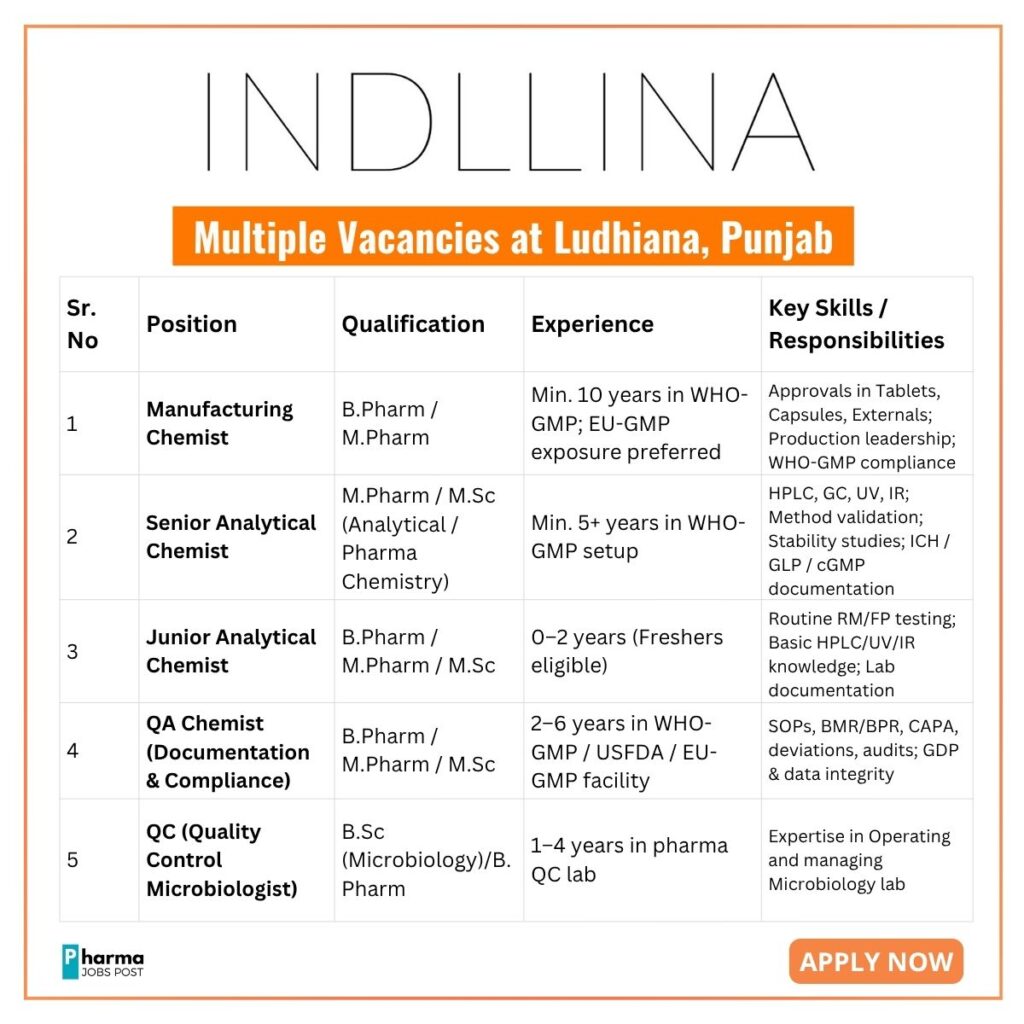
✅ Manufacturing Chemist
Qualification: B.Pharm / M.Pharm
Department: Production
Required Approvals:
- Tablets
- Capsules
- Externals (Topical Preparations)
Experience:
- Minimum 10 years in WHO-GMP certified plant
- Strong hands-on WHO-GMP compliance (mandatory)
- EU-GMP audit exposure preferred
Key Expertise:
- Regulatory & audit compliance
- Production leadership and multi-department coordination
✅ Senior Analytical Chemist
Qualification: M.Pharm / M.Sc (Analytical / Pharmaceutical Chemistry)
Department: Quality Control
Experience:
- Min. 5+ years in WHO-GMP environment
- Excellent command over HPLC, GC, UV, IR
- Strong knowledge of analytical method validation & ICH guidelines
Key Responsibilities:
- Conduct & review analytical testing
- Develop and validate analytical methods
- Train junior chemists
- Ensure cGMP & GLP compliance
✅ Junior Analytical Chemist
Qualification: B.Pharm / M.Pharm / M.Sc
Department: Quality Control
Experience:
- 0–2 years (freshers can apply)
- Basic understanding of HPLC / UV / IR preferred
Key Responsibilities:
- Routine raw material & finished product testing
- Support senior chemists in method development
- Maintain laboratory documentation & data integrity
✅ QA Chemist (Documentation & Compliance)
Qualification: B.Pharm / M.Pharm / M.Sc
Experience: 2–6 years in WHO-GMP / USFDA / EU-GMP plant
Responsibilities:
- SOP, BMR, BPR preparation & control
- CAPA, deviation, change control handling
- Support internal & regulatory audits
- Ensure GDP, data integrity & compliance
- Batch release coordination & validation documentation
✅ QC Microbiologist
Qualification: B.Sc (Microbiology) / B.Pharm
Experience: 1–4 years in pharmaceutical QC microbiology lab
Responsibilities:
- Microbial testing of water, RM & FP
- Environmental monitoring
- Sterility testing & microbial limits
- Operation & management of microbiology lab
- Maintain GLP, cGMP & data integrity
📧 How to Apply
Send your CV to:
Email: DAKSH@INDLLINA.COM
daksh@indllina.com
Subject Line:
Application – [Position Name] – Indllina Pharmaceuticals
Why Join Indllina Pharmaceuticals?
- WHO-GMP certified organization
- Compliance-driven excellence
- Hands-on technical and quality career growth
- Modern manufacturing setup with regulatory focus

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

