V-Ensure Pharma Technologies Announces Engineering Vacancies at Panvel Facility
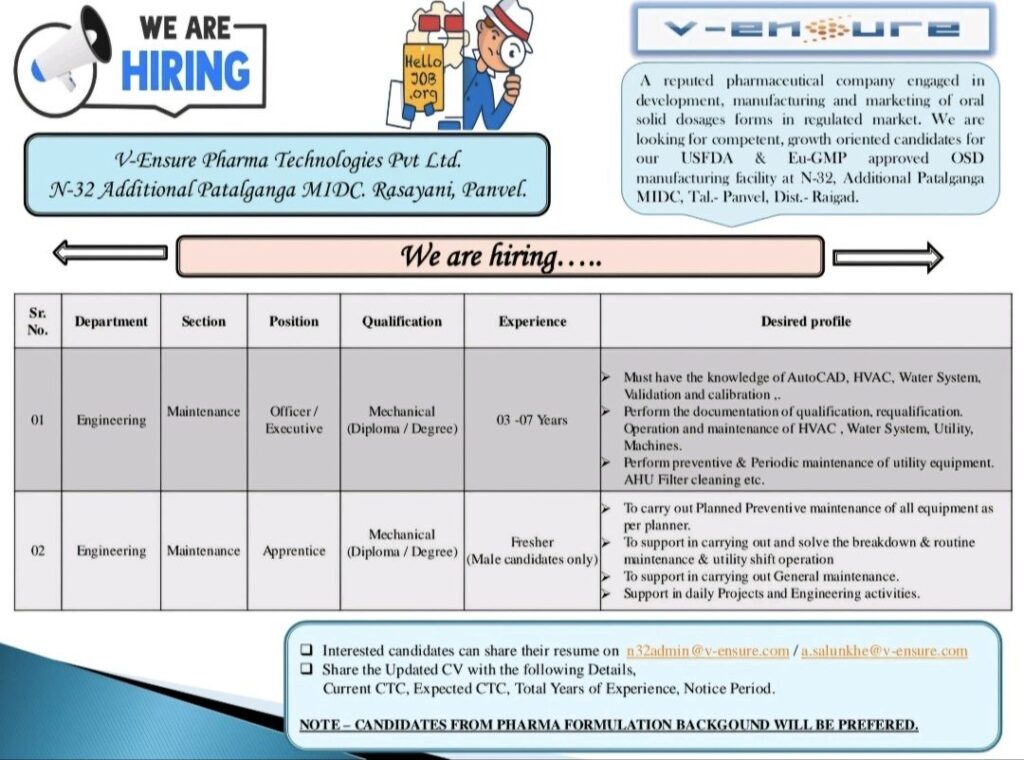
V-Ensure Pharma Technologies Pvt. Ltd., a reputed pharmaceutical company specializing in solid dosage forms for regulated markets, has announced openings in its Engineering Department at its USFDA and EU-GMP approved OSD manufacturing facility located in Additional Patalganga MIDC, Rasayani, Panvel.
About V-Ensure Pharma Technologies
V-Ensure Pharma Technologies is actively engaged in the development, manufacturing, and marketing of solid dosage forms in regulated markets. The company operates from its state-of-the-art facility at N-32, Additional Patalganga MIDC, Tal.- Panvel, Dist.- Raigad, which holds approvals from both USFDA and EU-GMP regulatory bodies.
Current Openings
The company is seeking competent, growth-oriented candidates for two positions in the Engineering Department’s Maintenance Section:
Position 1: Officer/Executive – Maintenance
Qualifications: Mechanical Engineering (Diploma/Degree)
Experience Required: 3-7 Years
Key Responsibilities:
– Proficiency in AutoCAD, HVAC, and Water System operations
– Expertise in validation and calibration processes
– Documentation of qualification and requalification procedures
– Operation and maintenance of HVAC, Water System, Utility, and Machines
– Preventive and periodic maintenance of utility equipment
– AHU filter cleaning and related maintenance activities
Position 2: Apprentice – Maintenance
Qualifications: Mechanical Engineering (Diploma/Degree)
Experience Required: Fresher (Male candidates only)
Key Responsibilities:
– Execute planned preventive maintenance of all equipment according to schedule
– Support breakdown resolution and routine maintenance during utility shift operations
– Assist in general maintenance activities
– Participate in daily projects and engineering activities
Application Process
Interested candidates can submit their resumes to:
Email: n32admin@v-ensure.com / a.salunkhe@v-ensure.com
Required Information:
– Updated CV with complete details
– Current CTC
– Expected CTC
– Total years of experience
– Notice period
Special Note: Candidates from pharma formulation backgrounds will be preferred for these positions.
Why Join V-Ensure Pharma Technologies?
Working at V-Ensure Pharma Technologies offers professionals the opportunity to be part of a company committed to quality manufacturing standards for regulated markets. The company’s USFDA and EU-GMP approvals demonstrate its commitment to maintaining the highest quality standards in pharmaceutical manufacturing.
FAQ:
1. What types of HVAC systems are commonly used in pharma OSD manufacturing facilities?
Pharmaceutical OSD units typically use centralized HVAC systems with AHUs, HEPA filters, dehumidifiers, and differential pressure control. These systems maintain controlled temperature, RH, and particulate levels essential for cGMP compliance.
2. What are the key validation activities involved in the maintenance of water systems?
Validation for pharmaceutical water systems includes:
IQ/OQ/PQ documentation
Conductivity, TOC, and microbial testing
Sanitization cycle verification
Flow rate, loop velocity, and dead-leg compliance checks
Regular calibration of sensors and instruments
3. How is preventive maintenance (PM) scheduled for utility equipment in a pharma environment?
PM is scheduled based on:
Manufacturer recommendations
Machine run-hours
Regulatory guidelines (USFDA/EU-GMP)
Criticality ranking of equipment (HVAC, water systems, compressors, boilers, etc.)
PM includes lubrication, alignment checks, replacements, calibration, filter cleaning, and performance testing.
4. What documentation is required for qualification and requalification of equipment in the maintenance department?
Standard documentation includes:
URS, DQ, IQ, OQ, and PQ protocols
Calibration certificates
Change control forms
Preventive maintenance records
Equipment history cards/logbooks
Requalification summary reports
5. What are common troubleshooting steps for breakdowns in HVAC or water systems?
Troubleshooting involves:
Checking differential pressure and airflow in AHUs
Inspecting filters (pre, fine, HEPA)
Verifying pump operation, flow rate, and valve positions
Checking electrical parameters: voltage, current, overloads
Reviewing BMS/SCADA alarms
Inspecting for leaks, blockages, or sensor failures

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

