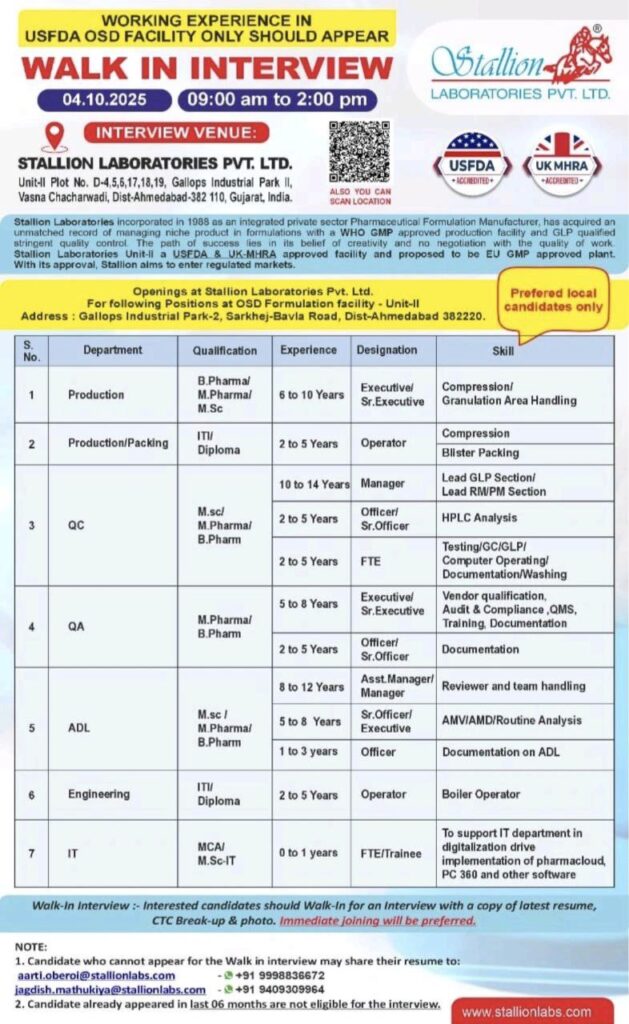
Stallion Laboratories Pvt. Ltd., a reputed pharmaceutical formulation manufacturer with USFDA & UK-MHRA approved facilities, is conducting a Walk-In Interview for experienced professionals at their OSD Formulation Facility. This is an excellent opportunity for candidates with relevant experience in USFDA-regulated plants to join a growing organization known for quality and compliance.
🗓 Interview Details
- 📅 Date: 4th October 2025
- ⏰ Time: 09:00 AM to 02:00 PM
- 🏢 Venue: Stallion Laboratories Pvt. Ltd.,
Unit-II, Plot No. D-4,5,17,18,19, Gallops Industrial Park II,
Vasna Chacharwadi, Dist-Ahmedabad – 382110, Gujarat, India. - 📍 Location: Gallops Industrial Park-2, Sarkhej-Bavla Road, Dist-Ahmedabad – 382220.
- 🔸 Preferred: Local candidates only
Current Openings
Production, Production/Packing, Quality Control (QC), Quality Assurance (QA), Analytical Development Laboratory (ADL), Engineering, Information Technology (IT)
| S. No. | Department | Qualification | Experience | Designation | Skills / Area |
|---|---|---|---|---|---|
| 1 | Production | B.Pharma / M.Pharma / M.Sc | 6–10 Years | Executive / Sr. Executive | Compression / Granulation Area Handling |
| 2 | Production / Packing | ITI / Diploma | 2–5 Years | Operator | Compression / Blister Packing |
| 3 | QC | M.Sc / M.Pharma / B.Pharma | 2–5 Years | Officer / Sr. Officer | HPLC Analysis |
| 2–5 Years | FTE | Testing, GC/GLP, Documentation, Washing | |||
| 10–14 Years | Manager | Lead GLP Section / Lead RM/PM Section | |||
| 4 | QA | M.Pharma / B.Pharma | 5–8 Years | Executive / Sr. Executive | Vendor Qualification, QMS, Training, Documentation |
| 2–5 Years | Officer / Sr. Officer | Documentation | |||
| 8–12 Years | Asst. Manager / Manager | Reviewer & Team Handling | |||
| 5 | ADL | M.Sc / M.Pharma / B.Pharma | 5–8 Years | Sr. Officer / Executive | AMV / AMD / Routine Analysis |
| 1–3 Years | Officer | Documentation | |||
| 6 | Engineering | ITI / Diploma | 2–5 Years | Operator | Boiler Operator |
| 7 | IT | MCA / M.Sc-IT | 0–1 Year | FTE / Trainee | Digitalization support, software implementation |
Additional Information
- Candidates with working experience in USFDA OSD facilities only should apply.
- Immediate joining will be preferred.
- Candidates who have appeared for an interview in the last 6 months are not eligible.
- Bring a copy of your latest resume, CTC breakup, and photograph when attending.
For Those Who Cannot Attend
Eligible candidates who are unable to attend the walk-in may send their resumes to:
- 📧 aarti.oberoi@stallionlabs.com
- 📧 jagdish.mathukiya@stallionlabs.com
- 📞 +91 9998836672, +91 9490399964
About Stallion Laboratories Pvt. Ltd.
Incorporated in 1988, Stallion Laboratories Pvt. Ltd. is a leading pharmaceutical formulation manufacturer with a WHO-GMP approved facility. The company has earned recognition for its stringent quality standards, creativity, and commitment to compliance. Stallion’s Unit-II is a USFDA & UK-MHRA approved facility and proposed to be EU GMP certified, making it a trusted name in regulated markets.
Don’t miss this opportunity to work with a leading pharma company in Ahmedabad.
Mark your calendar for 4th October 2025 and walk in for the interview.

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

