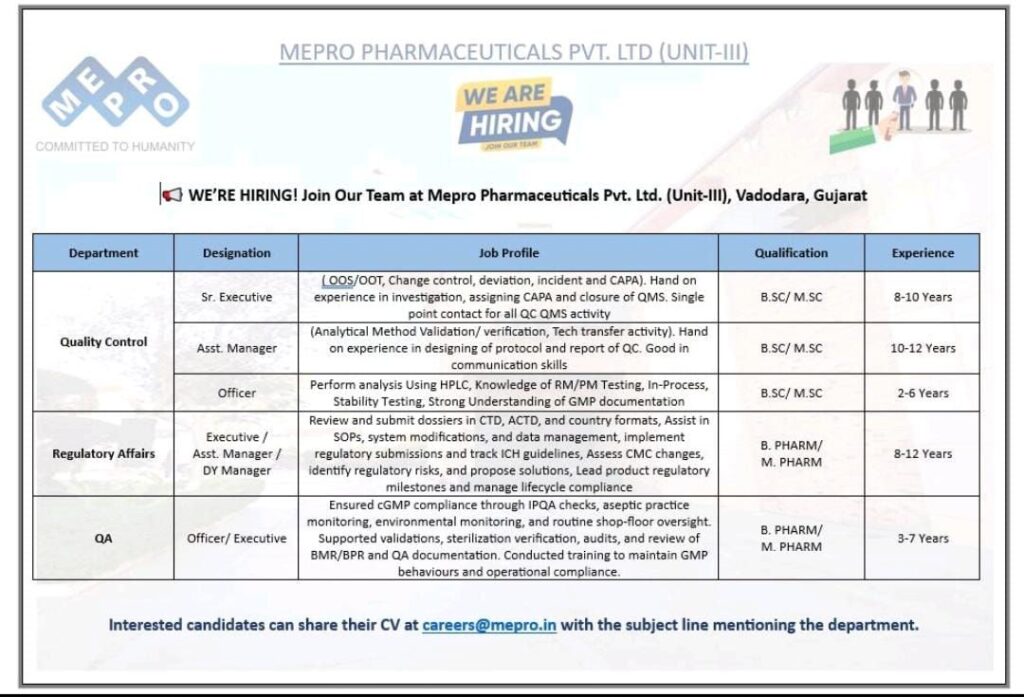
Mepro Pharmaceuticals Pvt. Ltd. (Unit-III) is expanding its team and actively recruiting experienced professionals for its facility in Vadodara, Gujarat. This is an excellent opportunity to advance your career in Quality Control (QC), Regulatory Affairs, and Quality Assurance (QA).
Quality Control (QC) Vacancies
Mepro is looking for dedicated individuals with a B.Sc./M.Sc. background to handle critical quality management tasks.
- Sr. Executive (Experience: 8-10 Years):
- Job Profile: Requires hands-on experience in the investigation, assigning CAPA (Corrective and Preventive Action), and closure of OOS (Out-of-Specification) / OOT (Out-of-Trend), Change Control, Deviation, and Incident. This role serves as a single point of contact for all QC QMS activity.
- Asst. Manager (Experience: 10-12 Years):
- Job Profile: Must have hands-on experience in designing of protocol and report of QC, specifically covering Analytical Method Validation/Verification and Technology Transfer activities. Strong communication skills are essential.
- Officer (Experience: 2-6 Years):
- Job Profile: Responsible for performing analysis using HPLC, possessing knowledge of RM/PM Testing, In-Process, and Stability Testing. A solid understanding of GMP documentation is required.
Regulatory Affairs (RA) & Quality Assurance (QA)
Opportunities are available for candidates with B. Pharm/M. Pharm qualifications to ensure compliance and support operational excellence.
Regulatory Affairs
- Executive/Asst. Manager/Dy Manager (Experience: 8-12 Years):
- Job Profile: Key responsibilities include reviewing and submitting dossiers in CTD, ACTD, and country formats. The role involves assisting in SOPs, system modifications, and data management, implementing regulatory submissions, and tracking ICH guidelines. You will assess CMC changes, identify regulatory risks, and propose solutions to manage product regulatory milestones and lifecycle compliance.
Quality Assurance (QA)
- Officer/Executive (Experience: 3-7 Years):
- Job Profile: Responsibilities include ensuring cGMP compliance through IPQA (In-Process Quality Assurance) checks, overseeing aseptic practice monitoring, conducting environmental monitoring and routine shop-floor oversight. This position supports validations, sterilization verification, and performs audits and review of BMR/BPR and QA documentation. The candidate will be trained to maintain GMP behaviours and operational compliance.
How to Apply
Interested and eligible candidates are invited to share their updated CVs with the Mepro team.
- Email: careers@mepro.in
- Subject Line: MUST mention the department you are applying for (e.g., QC, Regulatory Affairs, or QA).
Take the next step in your pharma career with Mepro Pharmaceuticals!

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

