Grow Your Career in Production and Quality Control at Lupin Ltd.
Lupin Limited is conducting a Walk-in Interview drive for experienced candidates to join their team at the Dabhasa facility, with interviews taking place in Ankleshwar. This is a prime opportunity for those with experience in the Pharma Industry, specifically in Production and Quality Control roles.
Contents
Production Department Opportunities
Lupin is hiring for two key production roles, focusing on API/Bulk Drug manufacturing:
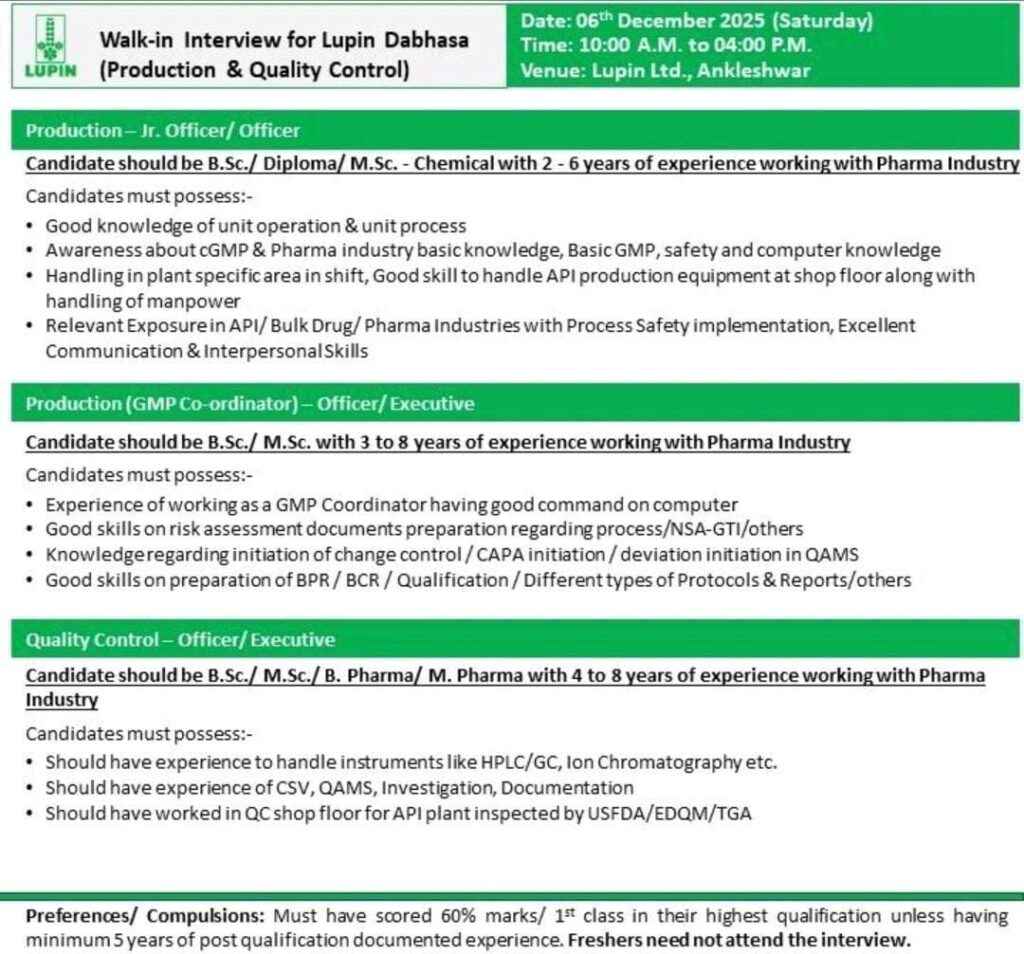
1. Production – Jr. Officer / Officer
- Experience Required: 2 to 6 years of experience working in the Pharma Industry.
- Qualification: B.Sc., Diploma, or M.Sc. in Chemical.
- Key Requirements:
- Good knowledge of unit operation & unit process.
- Basic knowledge of cGMP, GMP, safety, and computers.
- Ability to handle plant-specific areas in shift, managing API production equipment at the shop floor, and supervising manpower.
- Relevant exposure in API/Bulk Drug manufacturing with process safety implementation.
- Excellent communication and interpersonal skills.
2. Production – GMP Co-ordinator (Officer / Executive)
- Experience Required: 3 to 8 years of experience working in the Pharma Industry.
- Qualification: B.Sc. or M.Sc.
- Key Requirements:
- Experience working as a GMP Coordinator with good command on the computer.
- Good skills in risk assessment document preparation regarding Process/NSA-GTI/others.
- Knowledge of initiating change control, CAPA initiation, or deviation initiation in QAMS.
- Good skills in the preparation of BPR/BCR/Qualification and various types of Protocols & Reports/others.
Quality Control Department Opportunity
Lupin is seeking experienced professionals to ensure the highest standards of quality:
Quality Control – Officer / Executive
- Experience Required: 4 to 8 years of experience working in the Pharma Industry.
- Qualification: B.Sc., M.Sc., B. Pharm, or M. Pharm.
- Key Requirements:
- Should have experience handling instruments like HPLC/GC, Ion Chromatography, etc.
- Should have experience with CSV, QAMS, Investigation, and Documentation.
- Should have worked in a QC shop floor for an API plant inspected by regulatory bodies like USFDA/EDQM/TGA.
Interview & Mandatory Criteria
Interview Details
- Date: 06th December 2025 (Saturday)
- Time: 10:00 A.M. to 04:00 P.M.
- Venue: Lupin Ltd., Ankleshwar
Mandatory Preference/Compulsion
- All candidates must have scored 60% marks/1st class in their highest qualification.
- A minimum of 5 years of post-qualification documented experience is required.
- Freshers need not attend the interview.

 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

