Ipca Laboratories Walk-In Interview 2026 | Multiple Openings in Production, QA, QC & Packing
Ipca Laboratories Limited is conducting a walk-in interview for multiple departments including Production, Packing, Quality Assurance, Quality Control, and AMV. This is a great opportunity for experienced pharma professionals looking to grow their careers in a reputed organization.
Contents
📌 Job Details
- Company: Ipca Laboratories Limited
- Interview Date: 22 March 2026
- Timing: 9:30 AM – 5:00 PM
- Experience Required: 3 to 9 Years
- Job Location: Indore
📍 Walk-In Venue
Hotel Papaya Tree, Rau By-pass Square, Indore
Open Positions
- Production
- Packing
- Quality Assurance (QA)
- Quality Control (QC)
- AMV (Analytical Method Validation)
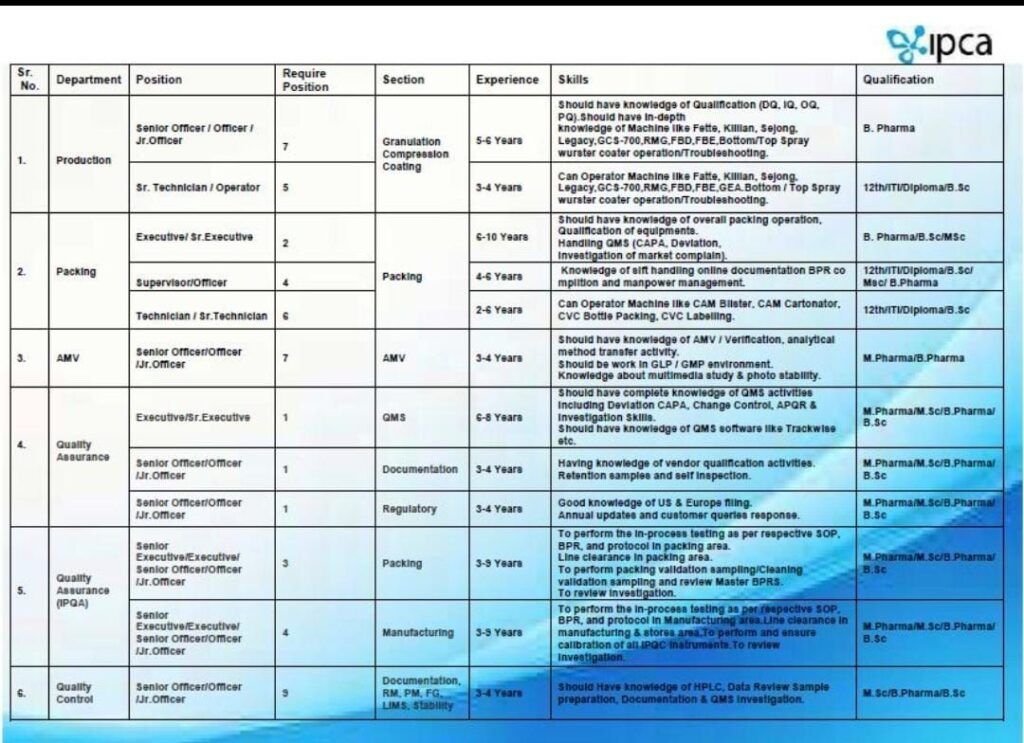
🧾 Department-wise Roles & Requirements
1. Production
- Positions: Officer / Jr. Officer / Sr. Technician / Operator
- Experience: 3–6 Years
- Qualification: B.Pharm / Diploma / B.Sc
- Skills: Granulation, Compression, Coating, handling equipment like Fette, Killian, Sejong
2. Packing
- Positions: Executive / Sr. Executive / Supervisor / Technician
- Experience: 2–10 Years
- Qualification: B.Pharm / B.Sc / M.Sc / Diploma
- Skills: Blister, Cartonator, documentation (BPR), packaging operations
3. AMV
- Positions: Officer / Jr. Officer
- Experience: 3–4 Years
- Qualification: B.Pharm / M.Pharm
- Skills: Method validation, GLP, documentation, stability studies
4. Quality Assurance (QA)
- Positions: Executive / Officer
- Experience: 3–8 Years
- Qualification: B.Pharm / M.Pharm / M.Sc
- Skills: QMS, CAPA, deviation handling, audit compliance, documentation
5. Quality Control (QC)
- Positions: Officer / Jr. Officer
- Experience: 3–4 Years
- Qualification: B.Sc / B.Pharm / M.Sc
- Skills: HPLC, data review, sample preparation, stability studies
🎓 Eligibility Criteria
- Candidates must have relevant pharmaceutical experience
- Strong knowledge of GMP, GLP, and documentation practices
- Experience in regulatory compliance is preferred
Important Note
Ipca Laboratories does not charge any fees for recruitment. Candidates are advised to be cautious of fraudulent job offers.
📢 How to Apply
Interested candidates can directly attend the walk-in interview at the venue with:
- Updated Resume
- Educational Certificates
- Experience Documents


 India's DMPLOI - Jobs & Networking App
India's DMPLOI - Jobs & Networking App

